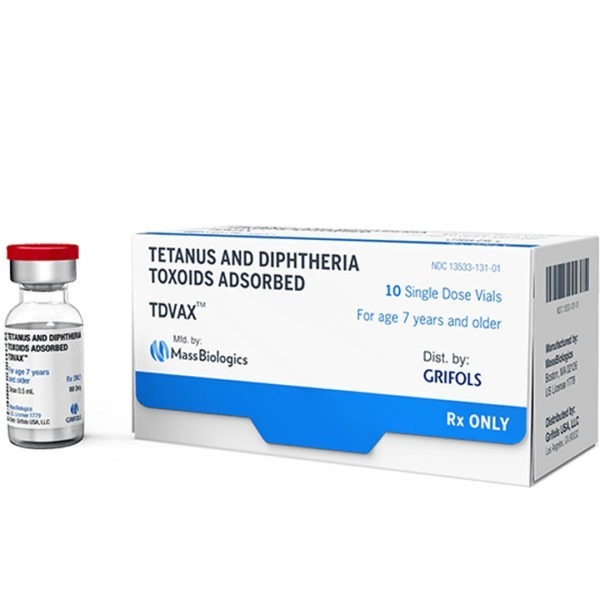
TDVAX Tetanus Toxoid/ Diphtheria Injectable SDV 0.5mL 10/PK
As Low As: $377.01 eachList Price: $471.26
- Description
- Product Spec
Description
TDVAXTM (tetanus and diphtheria toxoids adsorbed) is a vaccine indicated for active immunization for the prevention of tetanus and diphtheria. This vaccine is approved for use in persons 7 years of age and older.
- Manufacturer #: 13533013101
- Manufacturer: Grifols Therapeutics
- Country of Origin: United States
- Application: Td Vaccine
- Container Type: Single Dose Vial
- Dosage Form: Injection
- Generic Drug Code: 20835
- Generic Drug Name: Tetanus and Diphtheria Toxoids Adsorbed
- NDC Number: 13533-0131-01
- Storage Requirements: Requires Refrigeration
- Strength: 2 Lf – 2 Lf / 0.5 mL
- Type: Intramuscular
- UNSPSC Code: 51201629
- User: 7 Years of Age and Older
- Volume: 0.5 mL
TDVAX™ manufactured by MassBiologics is a sterile vaccine for intramuscular injection. After shaking, the vaccine appears as a homogeneous milky white suspension. Each 0.5 mL dose of MassBiologics’ TDVAX is formulated to contain the following active ingredients: 2 Lf of tetanus toxoid and 2 Lf of diphtheria toxoid. Each 0.5 mL dose also contains aluminum adjuvant (not more than 0.53 mg aluminum by assay), < 100 mcg (0.02%) of residual formaldehyde, and a trace amount of thimerosal [mercury derivative, (< 0.3 mcg mercury/dose)] (not as a preservative) from the manufacturing process. The Corynebacterium diphtheriae and Clostridium tetani organisms are grown on modified Mueller’s media1, 2 which contains bovine extracts. The bovine material used in these extracts is sourced from countries which the United States Department of Agriculture has determined neither have nor present an undue risk for bovine spongiform encephalopathy. Tetanus and diphtheria toxins produced during growth of the cultures are detoxified with formaldehyde. The detoxified materials are then separately purified by ammonium sulfate fractionation. The diphtheria toxoid is further purified by column chromatography. The tetanus and diphtheria toxoids are individually adsorbed onto aluminum phosphate. The tetanus and diphtheria toxoids induce at least 2 units and 1 unit of antitoxin per mL of serum, respectively, in the guinea pig potency test.
Additional information
| Weight | 1 lbs |
|---|---|
| Dimensions | 9 × 7 × 4 in |
| Brand | Generic |
| Product Category | |
| Quantity |