Home New
-
17 Panel Drug Test Cup
THC, COC, AMP, OPI, MAMP, BAR, BZO, OXY, MDMA, MTD, PCP, BUP, ETG, FEN, TRA, KRA, K2
As Low As:
3 Adulterants (PH/SG/CREAT)
$2.79 each Add to cartInstant 12-Panel Multi Drug Test Cup with PCP & 3 Adulterants
THC, COC, AMP, OPI300, MAMP, PCP, BAR, BZO, MTD, MDMA, OXY, BUP
As Low As:
3 Adulterants (PH/SG/CREAT)
$1.65 each Add to cart6 Panel Drug Test Cup with PCP & THC
MOP(OPI)300, PCP25, COC150, MET500, AMP500, THC50
As Low As:
$1.36 each Add to cart17 Panel Drug Test Cup with PCP, OXY & ETG
OPI300, MDMA500, OXY100, PCP25, COC150, MET500, MTD300, BAR300, BZO300, AMP500, PPX300, FYL10 TCA1000, EDDP300, THC50, BUP10, ETG500
As Low As:
$2.90 each Add to cart14 MultiPanel Test Cup with EDDP & 3 Adulterants
MOP(OPI)300, MDMA500, OXY100, PCP25, COC150, MET500, MTD300, BAR300, BZO300, AMP500, TCA1000, EDDP300, THC50, BUP10
As Low As:
3 Adulterants (PH/SG/CREAT)
$1.83 each Add to cart10-Panel Instant Multi Drug Test Cup with OXY & THC
MOP(OPI)300, OXY100, COC150, MET500, MTD300, BAR300, BZO300, AMP500, THC50, BUP10
As Low As:
$1.49 each Add to cartInstant Drug Testing Kits
Drug Test Cups
Drug Test Dip Cards
Saliva Drug Test Kit
Featured Products
-
Unit: CS25
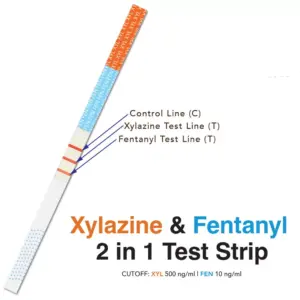
DOA Single Test Strip – Xylazine(XYL) 500ng/mL and Fentanyl(FEN)10ng/mL – Combo Single Test
WXY-FT-ST25 As Low As: $2.47 eachList Price: $81.56 -
Unit: C400
90mL (3 oz.) 53mm Specimen Cup, Screw on Teal Cap, Generic Label, Sterile w/ Temp Strip, Case of 400
39405MC As Low As: $0.36 eachList Price: $204.00 -
Unit: CS25
10-Panel Drug Screen Cup CLIA Waived – Precision DX – THC/COC/AMP/OPI/MAMP/BZO/MTD/MDMA/OXY/BUP
PREDX-CUP8104N As Low As: $2.49 eachList Price: $88.75 -
-
Unit: CS25
12-Panel Multi Drug Screen (THC/COC/AMP/MAMP/OPI/PCP/BAR/BZO/OXY/TCA/PPX/MTD) Dip Card – Precision DX – Expires April-2024
PREDX-DOA-1125NX As Low As: $0.75 eachList Price: $18.75 -
Unit: EACH
90 mL Specimen Cup, Screw on Cap, Generic Label, Sterile W/ Temp Strip (ASC-100)
39405A As Low As: $0.61 eachList Price: $0.63 -
Unit: EACH
Discover Lateral Flow (LF) Reader +
ATR-300 As Low As: $1,999.00 eachList Price: $1,999.00 -
Unit: EACH
Precision Reader w/ attachments (3-4 Week Lead Time)
DOA-ADT3000 As Low As: $3,300.00 eachList Price: $3,300.00 -
Unit: CS25
14-Panel Instant Drug Screen Cup – G-Cup – THC/COC/OPI/MAMP/AMP/FEN/MOP/OXY/MTD/BAR/BZO/BUP/K2/GAB
GCUP-2144N As Low As: $4.73 eachList Price: $168.75 -
Unit: CS25
19 Panel Drug Test Cup – W/ADS (6MAM/AMP500/BARB/BUP/BZO/COC150/FEN/K220/KET/KRA/MAMP500/MDMA/MOR/MTD/OXY/PCP/THC20/TRA/ETG) – (PH-SG-OX)
G-CUP-9191 As Low As: $6.39 eachList Price: $199.69
u003cspanu003eHEALTHY NEWS BLOGu003c/spanu003e
Drug Testing For Small Businesses
Any concern, whether big or small should adopt the policy of Drug Free Work Place. But, The National Drug-Free Workplace Alliance indicates that small businesses are the most affected compared to large businesses because of drugs and substance abusers. One of the reasons is drug abusers do not prefer large…
Difference between Drug Test Cups and Drug Test Dip Cards
Drug testing is a common occurrence in work places, educational institutions, law enforcing agencies and health care centres to screen for illicit drugs in order to maintain safety. Different types of devices and tools are available to detect candidates for substances of abuse and drugs. Drug Test Cups and Drug…
Mouth Swab Drug Test: An Overview of Saliva Drug Test
Due to the priority in maintaining safety at work places or institutions, drug screening has become very necessary and inevitable. A Mouth Swab Drug Test is a less invasive screening technique, which uses the saliva sample of an individual to check for the consumption of any illegal drug. Though different…
HEALTHY NEWS BLOG
Drug Testing For Small Businesses
Any concern, whether big or small should adopt the policy of Drug Free Work Place. But, The National Drug-Free Workplace Alliance indicates that small businesses are the most affected compared to large businesses because of drugs and substance abusers. One of the reasons is drug abusers do not prefer large…
Difference between Drug Test Cups and Drug Test Dip Cards
Drug testing is a common occurrence in work places, educational institutions, law enforcing agencies and health care centres to screen for illicit drugs in order to maintain safety. Different types of devices and tools are available to detect candidates for substances of abuse and drugs. Drug Test Cups and Drug…
Mouth Swab Drug Test: An Overview of Saliva Drug Test
Due to the priority in maintaining safety at work places or institutions, drug screening has become very necessary and inevitable. A Mouth Swab Drug Test is a less invasive screening technique, which uses the saliva sample of an individual to check for the consumption of any illegal drug. Though different…
Clearance